In short
- Ultrasound is increasingly studied as a method to control algae blooms, a global problem that worsens each year;
- Ultrasonic treatment has the power to effectively control these harmful blooms;
- Not all ultrasonic algae control technologies are the same.

In recent years, various innovations regarding lake restoration and algal bloom remediation have been introduced and tested. For example, One algae bloom control method is using interactive ultrasound programs that control algae growth.
Therefore, with every new technology, researchers must study its effects on algae and the surrounding ecosystem. This blog addresses the most common concerns regarding the use of ultrasound in lakes/reservoirs based on research conducted by various academic institutions.
Ultrasound research developed by European universities

The Chameleon Technology™ for algae control, also known as Monitor-Predict-Control (MPC) technology, was developed based on a large research grant from the European Commission[1]. The technology resulted from the collaboration between:
- The KAUNO Technological University;
- The Hellenic Centre for Marine Research;
- The Technological Institute of Norway;
- The Meteorological Institute of Poland, and
- a group of industry partners.
As a result, these studies produced the MPC-Buoy technology, a solar-powered buoy that uses in-situ, real-time water quality monitoring to identify and predict algal growth and autonomously applies specific ultrasonic frequencies to match the algal type present in the water.

To support this, researchers built a database that contains algal types, water quality data, and matching ultrasonic parameters.
This database operates an algorithm that drives ultrasound program changes in the MPC-Buoy. Over time, the system automatically updates this database with every new reading collected from MPC-Buoys operating worldwide.
Manipulating light to reduce cyanobacteria
When a water body suffers from harmful algal blooms, solutions are most often sought in the form of reducing phosphate – the key nutrient that algae and cyanobacteria consume to grow. However, aside from phosphate, light is also a critical resource for all phytoplankton.
The University of Missouri (MO) and the University of Kansas (KS) recently explored light manipulation as a feasible method to reduce cyanobacteria. They found that reducing photosynthetic active radiation (PAR) in a water body by 60.4–86.4% led to a 77.9% decline in cyanophyte biovolume in tanks dominated by cyanobacteria. As a result, limited light availability drove most of the change.
Additionally, these studies show that the approach of control of algal growth while keeping phosphate levels stable throughout the experiment.[2]
Similarly, LG Sonic’s MPC-Buoy works through a similar principle. Instead of limiting nutrients, it reduces the light availability to disrupt the growth of cyanobacteria and other blooming algae. Other studies, such as research by the University of Aarhus in Denmark, show that PAR declines exponentially with depth in both shallow and deeper water bodies. [3]
Therefore, instead of reducing light at the surface, ultrasound can move algal cells deeper, where PAR levels are lower.
Affecting algae’s buoyancy
The MPC-Buoy uses ultrasound as a mechanism to accomplish this. Ultrasound adds hydrostatic pressure around the algal cell, affecting the buoyancy of the cell. The algae require cell buoyancy to remain in the upper layer of the water and obtain sufficient light to bloom.
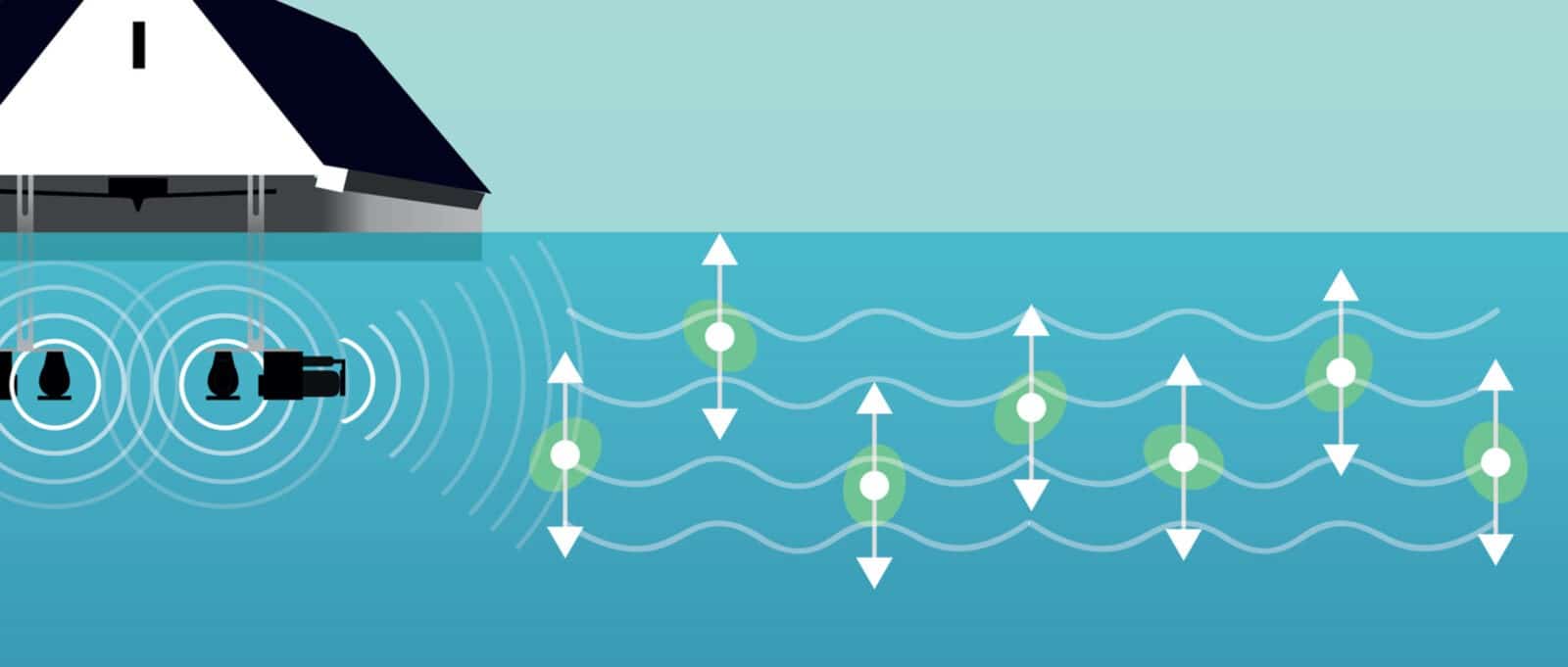
Many people think algal buoyancy depends on gas vesicle formation and collapse, but this explanation is only partly true. Collapse of gas vesicles is irreversible and for some algal types, not possible due to the size and shape of the vesicle.
In practice, the vertical migration of these algae is a more complex function. Gas vesicles provide lift that counteracts cell ballast such as carbohydrates and proteins. Algae regulate this balance based on water quality (turbidity, phosphate, and nitrate concentrations) [4], and hydrodynamic conditions such as hydrostatic pressure, waterflow and mixing of the surface column.
This is the area in which the ultrasound plays a key role, as the ultrasonic waves form a continuous pressure cycle around the algal cells, affecting the buoyancy regulation, which does not allow them to obtain sufficient buoyancy to remain the area where PAR is high enough for them to bloom.
This explains the need for frequent changes in ultrasonic parameters such as amplitude, frequency, and waveform. Buoyancy regulation is a continuous process closely linked to algal photosynthesis. It is, therefore, important that the hydrostatic pressures around the algae continuously change.
American Water’s research
For example, in 2014, Dr. Orren D. Schneider and Lauren A. Weinrich tested this theory. They performed in-situ tests with 4 MPC-Buoy systems in a drinking water reservoir in New Jersey for a period of 5 months.

The team took water samples throughout the test period and identified algal species. They found during the initial part of the study, algae were controlled well by the MPC-Buoys. However, on August 13, the team opened an inlet from an untreated reservoir because the plant needed additional water. This algae-laden water entered Reservoir 1 and seeded the reservoir with Aphanizomenon, a filamentous cyanobacterium.
The team detected increased algae, organic carbon, and taste- and odor molecules at the plant intake. Aphanizomenon started blooming in the reservoir and continued until the team changed the ultrasonic program to match this species. [5]
Why control algae if nutrients are the problem?
Algal blooms are often caused by eutrophication, a process in which nitrogen and phosphorous loads in a water body increase to a point that algae and plants start growing excessively. Once algae start blooming, it leads to increased turbidity, chlorophyll a, pH, and phycocyanin. These parameters have a negative effect on the growth-rate of submerged macrophytes and other aquatic organisms.
Submerged macrophytes play a key role in the clarity of the water within an ecosystem; first because they consume nutrients that would otherwise be available for algal growth, and also because submerged aquatic plants provide shelter for zooplankton against fish.
Zooplankton are natural phytoplankton grazers. When zooplankton disappear, water clarity declines as phytoplankton growth increases. Another important effect of reduced plant growth and water quality is on levels of Dissolved Oxygen (DO) within a water body, which can become unstable, and the deepest water levels (hypolimnion) can become anoxic (lack of oxygen). Thus, the microbial aerobic digestion in the deeper water layers of nutrients become compromised.
When algae blooms
As a result, the blooming of algae in a water body affect the entire production chain in water. In the hypolimnion, anoxic conditions lead to a reduced organic decomposition. This means that the sediment layer continues to grow. Due to lack of oxygen, it may release its content (nutrients, metals) into the water column, often leading to a further increase in algal proliferation.

As a result, under these conditions, reducing nutrient inflow will not alter any of the water quality parameters mentioned before. On the contrary, the sediment will continue to release nutrients as long as algae are blooming, and the water quality will further deteriorate.
Therefore, to actively solve the problem of algal blooms, it is important that algal proliferation is first controlled so the water quality can stabilize, allowing the aquatic ecosystem to adjust.
Ultrasonic effects on non-target organisms
Cavitation is another important concept of ultrasound waves. Cavitation occurs when ultrasound or other hydrostatic pressure in a liquid forms micro-bubbles that implode, creating intense heat, pressure, micro-jets, and hydrogen free radicals.
Industry uses cavitation in a wide variety of processes, such as cleaning jewelry, sterilizing surgical equipment, homogenizing samples, and lysing cells for laboratory applications.
Researchers often characterize it using high frequencies in the MHz range. Although the forms and characteristics of the cavitation may differ, it can be generated both at low (kHz) and high (MHz) frequencies, but primarily depends on output power.
Lowe & L. Brand [6] reflect in their paper on the energy requirement to generate cavitation in water, and Joyce et al. (2010)[7] cited cavitation as a primary cause of algae control within their experiment (power per volume range of 0.0015–0.17W/cm3).
Therefore, studies with a power per volume range above this threshold likely utilize cavitation. However, other experimental factors can also contribute to cavitation. LG Sonic products, generally uses power volumes of 0.55*E-8 and 0.33*E-6 respectively, which stay well below the cavitation threshold.
However, many scientific studies involving ultrasound have based their methods on technologies generating cavitation, which typically use low volumes of water and equipment with high amplitudes well within the range of generating cavitation.
As far as LG Sonic is concerned, these applications would damage non-target organisms as well as algae, but could not be upscaled to a real environment, as it would require too much energy and consume ultrasonic transducers.
Effects of ultrasound on fish
De Lange (2007) conducted a study on ultrasonic systems used for algae control in surface waters and their effects on several fish species: bream, bass, common roach (Rutilus rutilus), silver bream, ruffle (Gymnocephalus cernua), common rudd (Scardinius erythrophthalmus), tenches (Tinca tinca), and pikes (Esox lucius).
Researchers connected two identical basins stocked with fish through a tunnel so fish could migrate between them. De Lange used fish migration, as well as the size of the fish, as parameters to indicate stress or other effects of the ultrasonic systems.

The team sampled the fish population before activating ultrasound, after one month, and after four months. Results showed an even fish distribution across both basins after four months of ultrasound operation. This suggests ultrasound does not affect fish migration between the basins. The length of the fish between basins did not show any significant differences either.
De Lange also did not find any increased fish mortality in the experiment; therefore, researchers concluded that the ultrasonic system was neither noticeable nor unsafe for the tested fish species.[8]
Griessler Bulc et al. (2011) studied a water treatment system for common carp (Cyprinus carpio) which included ultrasound. They used equipment produced by LG Sonic for their tests, which was performed in 2 fish ponds: one with the ultrasonic unit and the other without. They concluded that with treatment (including ultrasound), rearing conditions for common carp were better with higher body weight increase and higher fish production than in the pond without treatment. [9]

Krivograd Klemenčič & Griessler Bulc performed more tests on aquaculture with ultrasonic systems in 2013 [10] and 2015 [11], both on lab scale and in a fish pond. Both tests did not show any adverse effects on the fish, while the study inside the fishpond did show higher fish yield compared to the untreated pond.
Techer et al. studied the effects of ultrasonic irradiation on fish in 2017 using a commercially available low-power ultrasonic generator. Their study focused on long-term effects on the common carp (Cyprinus carpio), using 2-year-old carp that were exposed to low-power ultrasound for 30 days. These results showed that carp were unaffected by ultrasound exposure when exposed in floating cages in fishponds over a 30-day period. [12]
Effects of ultrasound on zooplankton
Lab tests on Daphnia magna
The university of Ljubljana performed field and lab tests on Daphnia magna using equipment manufactured by LG Sonic. In the lab-scale tests, a set of adult and juvenile Daphnia magna were exposed to ultrasound and mobility was tested after 0, 4 and 19 hours of exposure.
Notably, in both tests, no significant differences were found in the mobility of the species after 19 hours, indicating that there was no acute effect of ultrasound exposure on the tested organisms.
Field tests and cage studies
In the field test, researchers placed the ultrasonic system in a pond and sampled zooplankton at 0, 12, 24, and 48 hours using a conical plankton net. They used two sampling sites: one in front of the ultrasonic system and one 7 meters away. The team analyzed the samples and determined total zooplankton numbers and taxa.
Researchers observed a slight increase in zooplankton close to the ultrasonic transmitter. However, researchers did not observe significant differences before sonication (t0) or at later time points, including after 48 h of sonication The differences are likely due to fish predation or movement of the zooplankton. [13]

In 2017, the aquatic research and consultancy Ecofide studied the effects of the MPC-Buoy systems, produced by LG Sonic. This study was commissioned by the Zoetermeer municipality and Rijnland Water Board to research the effects of the used ultrasonic technology on zooplankton.
For this study, researchers placed lab-bred zooplankton in cages for seven days. After seven days, researchers counted surviving water fleas and the number of young produced. This testing method is and has been applied often to field research regarding the effects on zooplankton.
For example, the Delfland Water Board has been using this method for years to determine the effects of pesticides (for example see Ecofide overview report 2008), as well as in the current Waternet research (Simoni, part of Ecological Key Factor toxicity); these field experiments form an important part (van der Oost et al., 2017; Stowa, 2016).
Researchers placed cages at different distances from the ultrasonic transmitters. The closest cages were placed under the buoy right in front of the transmitters, while the furthest transmitters were placed at the lake shore. Both the survival of the mature water fleas, as well as the reproduction (number of young) after 7 days, have been tested.
Researchers found no negative effects of ultrasonic sound waves on water flea survival, and average survival stayed at 95–100%. [14]
References
[1] The research leading to these results has received funding from the European Union Seventh framework Programme ([FP7/2007-2013] [FP7/2007-2011]) under grant agreement n° [FP7-SME-2011-286875-CLEARWATERPMPC].
[2] Gaskill JA, Harris TD and North RL (2020) Phytoplankton Community Response to Changes in Light: Can Glacial Rock Flour Be Used to Control Cyanobacterial Blooms? Front. Environ. Sci. 8:540607. doi: 10.3389/fenvs.2020.540607
[3] Gonçalves-Araujo R and Markager S (2020) Light in the Dark: Retrieving Underwater Irradiance in Shallow Eutrophic Waters From AC-S Measurements. Front. Mar. Sci. 7:343. View publication
[4] Zhaosheng Chu, Xiangcan Jin, Bo Yang, Qingru Zeng, Buoyancy regulation of Microcystis flos-aquae during phosphorus-limited and nitrogen-limited growth, Journal of Plankton Research, Volume 29, Issue 9, September 2007, Pages 739–745.
[5] Schneider, O.D., Weinrich, L.A. and Brezinski, S. (2015), Ultrasonic Treatment of Algae in a New Jersey Reservoir. Journal ‐ American Water Works Association, 107: E533-E542.
[6] Lowe M. 2011. Ultrasonic control of algae in stormwater systems. Water New Zealand, 7th South Pacific Stormwater Conference.
[7] Joyce, E. M., Wu, X., & Mason, T. J. (2010) ‘Effects of ultrasonic frequency and power on algae suspensions’ Journal of Environmental Science and Health, Part A, 4, 7, 863 – 866.
[8] De Lange M.C. 2007. Control of blue algae with ultrasound – the effects on fish. Report: VA2007_28. VisAdvies BV, Utrecht, 10 pp.
[9] Griessler Bulc T., Istenič D., Krivograd Klemenčič A. 2011. The efficiency of a closed-loop chemical-free water treatment system for cyprinid fish farms. Ecological engineering 37, 873-882.
[10] Krivograd Klemenčič A., Griessler Bulc T. 2013. The efficiency of constructed wetland and ultrasound for water reuse in a closed-loop small-scale cyprinid fish farm. Fresenius environmental bulletin, 22(10), 2828-2835.
[11] Krivograd Klemenčič A., Griessler Bulc T. 2015. The use of vertical constructed wetland and ultrasound in aquaponic systems. Environmental science and pollution research international, 22(2), 1420-1430
[12] Techer D., Milla S., Banas D. 2017. Sublethal effect assessment of a low-power and dual-frequency anti-cyanobacterial ultrasound device on the common carp (Cyprinus carpio): a field study. Environmental Science and Pollution Research 24(6), 5669–5678.
[13] Krivograd Klemenčič, Griessler Bulc, Pflieger. 2018. The effects of the LG Sonic technology on toxin degradation and on the environment (without algae). UNIVERSITY OF LJUBLJANA.
[14] Rineke Keijzers & Jaap Postma, Ecofide. Field research regarding possible effects of ultrasonic sound on zooplankton (2017)
